Fishing Effects on Fish Population Structure and Ecology
_Size-selective fishing will affect species with different life history
traits in different ways. Since species with late maturity and slow growth to a
larger size are typically the target species for the majority of fisheries, the
species composition of fish communities is expected to change. Smaller,
faster-growing species are projected to dominate the fish biomass. Moreover,
changes will be seen in fecundity, age and size structure, abundances of fish
populations, spawning potentials, sex ratios, and genetics within target
resources and their dependent species.
TABLE OF CONTENTS
Fecundity (or reproduction of numerous offspring) decreases are an important consequence of the way we fish due to the disappearance of the largest, most fecund individuals. Larger fish produce far more eggs than smaller fish as well as eggs with greater amounts of stored energy and growth hormones, providing larger larvae at hatching. Furthermore, bigger larvae have been revealed to show signs of greater feeding habits, the development of more functional swim bladders, as well as higher rates of swimming activity and growth. All these things combined will allow for better survival rates.
The loss of the most fecund individuals is well demonstrated amongst the Gag grouper populations in the Gulf of Mexico. Gag groupers are one of the most commercially important species in the western Atlantic, and can be found on menus all over the Florida coast. Female gags start reproducing when they reach age three or four and usually live to be about 30-35 years. During the time that they are reproductively active (more than a few decades) they can grow up to 51 inches long and weigh up to 55 pounds. The problem is that most Gags are typically between 2-5 years old when they are caught, and Gags older than 12 years of age are seldom even seen. Shortening the age structure of these fish means that the largest, most fecund fish no longer exist in fish populations.
_Age structure alterations are also a
significant result of fishing and apparent in Gag grouper communities.
Gags are hermaphrodites, changing from female to male when they reach a
certain size and age. This means the most desirable (older) fish are
typically males, causing decreases in populations when spawning
aggregations are targeted. This has resulted in a shift in the sex
ratio from 5 females to 1 male to 30 females to 1 male.
An alteration to age structure has also been seen amongst the endangered Shortnose Sturgeons. This species can be found in estuarine environments and along continental shelves. Although it has rarely been a target species, it has historically been a large bycatch of the commercial fishery of Atlantic sturgeon, and populations were well diminished in the late 1800s when dredging began altering habitat structures of this species. Males hardly ever live past the age of 30 while females live up to 67 years, resulting in a sex ratio of 1:1 for younger adults, but a ratio of 4:1 for mature adults, supporting the catch of females(larger) and implying the further decrease in overall sturgeon abundances.
An alteration to age structure has also been seen amongst the endangered Shortnose Sturgeons. This species can be found in estuarine environments and along continental shelves. Although it has rarely been a target species, it has historically been a large bycatch of the commercial fishery of Atlantic sturgeon, and populations were well diminished in the late 1800s when dredging began altering habitat structures of this species. Males hardly ever live past the age of 30 while females live up to 67 years, resulting in a sex ratio of 1:1 for younger adults, but a ratio of 4:1 for mature adults, supporting the catch of females(larger) and implying the further decrease in overall sturgeon abundances.
_Genetic changes in
life history traits may be a significant reason as to why fish
populations fail to recuperate even after considerable reductions in
fishing pressure. While fish growth is indeterminate, and naturally
occurring fish will continually grow with age, the desire to catch
larger and larger fish has also ended up removing the oldest and fastest
growing fish from populations. Fishing in such a manner has caused a
strong directional selection favoring the survival of younger, smaller,
and slower growing phenotypes. Under intense size-selective fishing,
natural selection has caused traits that would normally be favored under
natural conditions ( fast growth, high feeding rates, and large size)
to reduce fitness.
One example of this has been seen in the weight of Pacific salmon.
Since the 1950s, there has been a decline in the body weight of all five
species, resulting in the adult pink salmon to decrease its adult body
weight by up to 34%. The decrease in body weights is due to genetic
alterations following the 1940s, when fishermen began to increase the
mesh size of gillnets, making it more advantageous to be small in order
to escape being gill netted.
Effects of Fishing on Soft Sediments

_
Habitat structures such as coral
reefs, sea grasses, and soft sediments are vital in the organizing and
functioning of ecosystems for a variety of marine organisms including
commercially and recreationally valuable species. They provide a refuge from
predators, competition, and stresses, as well as areas for larvae recruitment. Habitats are also used as a place to spawn, nurse, and eat. Moreover, they influence
species biomass, richness, and composition.
A common view on marine seafloor biodiversity reflects a strong interest in hard bottoms such as coral reefs, kelp forests, and the rocky intertidal. However, about 70% of the earth's seafloor is composed of marine soft sediments. These soft-sediment habitats can be highly productive in allowing physical and biological interactions that support extremely high species diversity, abundance, and biomass.
A common view on marine seafloor biodiversity reflects a strong interest in hard bottoms such as coral reefs, kelp forests, and the rocky intertidal. However, about 70% of the earth's seafloor is composed of marine soft sediments. These soft-sediment habitats can be highly productive in allowing physical and biological interactions that support extremely high species diversity, abundance, and biomass.
_Grain-size composition, which is influenced by water depth, current speeds, and
organic content of the sediment, is a large determinant of where
macrofaunal species(larger than 1 mm in size)are distributed throughout
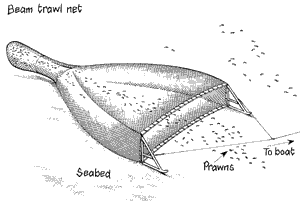
sediments. Bottom trawling causes disturbance to the seabed, interfering with
the physical and chemical properties of the habitat, leading to direct
mortality of benthic organisms. Depending on the grain size, the type of trawl
used, and the frequency of disturbance, the direct effects to soft sediment
organisms can be dire. Many species, especially invertebrates, are caught as
bycatch when fishermen drag the net along the bottom of the seabed. Most
species thrown back don’t survive and even if bottom dwelling species are not
caught directly by the nets, they can still be killed by the gear touching them
or by being exposed to predators.
In the year 2000, studies in the south-eastern North Sea found after just a single passage of a 4 m or 12 m wide trawl, there was 5%-50% mortality seen amongst invertebrate species and up to 68% for bivalve species, observing more mortality in the silty areas than in the sandy areas. This may be due to the depth in which the trawl penetrated into the sediment. This suggested that beam trawls break through silty sediment more deeply, leading to more mortalities. The time of year in which trawling is done may affect more or less organisms. For example, if one were to assume that the majority of the animals in reach of the trawl would die, mortality could increase up to 90% during their reproduction season when animals surface. The consequences of trawling in mud are even more surprising.
In the year 2000, studies in the south-eastern North Sea found after just a single passage of a 4 m or 12 m wide trawl, there was 5%-50% mortality seen amongst invertebrate species and up to 68% for bivalve species, observing more mortality in the silty areas than in the sandy areas. This may be due to the depth in which the trawl penetrated into the sediment. This suggested that beam trawls break through silty sediment more deeply, leading to more mortalities. The time of year in which trawling is done may affect more or less organisms. For example, if one were to assume that the majority of the animals in reach of the trawl would die, mortality could increase up to 90% during their reproduction season when animals surface. The consequences of trawling in mud are even more surprising.
Muddy sediments are perhaps the most prevalent of all seabed types, supporting a wide variety of important commercial fisheries as well as diverse and species rich communities. Consequently, muddy sediment communities are subject to regular fishing disturbance. Muddy sediments form in areas of rapid sedimentation, full of extensive burrows, where there is low-energy. Although muddy sediments protect species from most natural disturbances, faunal communities living here are greatly affected by even the smallest human disturbance. In contrast, species living in coarser sediments are subject to a greater level of natural disturbance, making the inhabitants more resilient to stress.
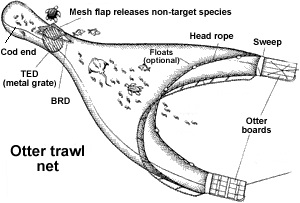
Trawls influence muddy sediments more than harder sediments because trawl doors can penetrate more deeply in mud (8-17 cm in the mud and 0-5 cm in the sand), creating trenches and flattening burrows . This results in potentially greater effects on infaunal species. Flattening of burrows may lead to a large degree of smothering as well as resuspension, causing larger areas of destruction and mortality. In addition, trawling has the tendency to remove larger and older aged species. Once these organisms are removed, it may take longer for the community to recover. The removing of the larger species may also lead to more mixing within the mud, altering fluxes between the water column and the sediment. This results in an increased chance of eutrophication and a longer time towards recovery. The rate in which such areas can recover is unclear, but seems to take more than a years’ time in muddy sediments in comparison to sand, where it takes 40 days to 6 months, depending on the type of trawl used. Otter trawls tend to catch less benthos and disturb a lesser depth than beam trawls.
_
The effects
of trawling can be apparent for long periods of time. For example, scientists
in the Scottish Sea observed that even 18 months after trawling had occurred,
trawl door tracks could still be detected by side-scan sonar in the muddy
seabeds.
Fishing disturbances alter macrobenthic community structure in muddy sediments differently than in harder substrates. A fishing experiment in Loch Gareloch over the course of 16 months showed that disturbance increased individual abundances, but reduced diversity. Echinoderms and large bivalves were hardly ever found at the heavily trawled sites and were instead conquered by carnivorous polychaetes and other opportunistic species.
Fishing disturbances alter macrobenthic community structure in muddy sediments differently than in harder substrates. A fishing experiment in Loch Gareloch over the course of 16 months showed that disturbance increased individual abundances, but reduced diversity. Echinoderms and large bivalves were hardly ever found at the heavily trawled sites and were instead conquered by carnivorous polychaetes and other opportunistic species.
Nutrient
levels can
be drastically altered by physical disturbances such as trawling. Sediment
resuspension occurs naturally in marine ecosystems by bioturbation, waves and
tidal currents, as well as sediment mixing by burrowing benthic organisms,
however trawling can cause increased levels of sediment buildup resulting in
vast alterations to the biogeochemistry of marine systems.
Nutrient input to sediment-inhabiting organisms is derived from the breaking down of decaying organic matter within sediments, followed by the upward movement by diffusion and biological advection. Trawling can affect sediment’s organic matter decay and nutrient fluxes several ways. One effect can be seen in the lowering of unstable organic matter from the sediment-water interface down into the subsurface layers. This can result in a shift from aerobic processes to anaerobic processes. The reverse effect would also be seen, resulting in the anaerobic layers becoming aerobic. The degree to which trawling induced mixing occurs and the depth and thickness of the RPD zone (Redox Potential Discontinuity)_ will determine the severity of affected populations that fix organic matter.
Another effect of bottom trawling on nutrient levels in soft sediments is the accelerating of nutrient inputs. The resuspension of just 1 mm of sediment has the capability of doubling or tripling the nutrient flux into the euphotic zone and can alter phytoplankton species and biomass dramatically, and therefore primary production.
Alterations to burrow spacing , abundance, and size can have detrimental effects to the fluxes of nitrogen between the water column and sediment surface. Nitrogen-fixing bacteria remove several tens of percentages of nitrogen before its distributed throughout the water column, by burrows and diffusion, to be used by plants and animals for chemical processes. Trawling may mix up the soil so much that ammonia will be introduced into the aerobic soil in mass amounts, helping to fuel the production of nitrate. Additionally, trawling may supply extra amounts of silica to the soil. These changes can be both good and bad for the ecosystem. Diatoms require silica to survive, so the extra input of silica into the soil could benefit the production of harmless diatoms which would help support primary production. However, toxic dinoflagellates (another species of phytoplankton) thrive on excess nitrogen, so the extra input of nitrogen into the water column may stimulate phytoplankton that could kill fish and other organisms living in the water.
Nutrient input to sediment-inhabiting organisms is derived from the breaking down of decaying organic matter within sediments, followed by the upward movement by diffusion and biological advection. Trawling can affect sediment’s organic matter decay and nutrient fluxes several ways. One effect can be seen in the lowering of unstable organic matter from the sediment-water interface down into the subsurface layers. This can result in a shift from aerobic processes to anaerobic processes. The reverse effect would also be seen, resulting in the anaerobic layers becoming aerobic. The degree to which trawling induced mixing occurs and the depth and thickness of the RPD zone (Redox Potential Discontinuity)_ will determine the severity of affected populations that fix organic matter.
Another effect of bottom trawling on nutrient levels in soft sediments is the accelerating of nutrient inputs. The resuspension of just 1 mm of sediment has the capability of doubling or tripling the nutrient flux into the euphotic zone and can alter phytoplankton species and biomass dramatically, and therefore primary production.
Alterations to burrow spacing , abundance, and size can have detrimental effects to the fluxes of nitrogen between the water column and sediment surface. Nitrogen-fixing bacteria remove several tens of percentages of nitrogen before its distributed throughout the water column, by burrows and diffusion, to be used by plants and animals for chemical processes. Trawling may mix up the soil so much that ammonia will be introduced into the aerobic soil in mass amounts, helping to fuel the production of nitrate. Additionally, trawling may supply extra amounts of silica to the soil. These changes can be both good and bad for the ecosystem. Diatoms require silica to survive, so the extra input of silica into the soil could benefit the production of harmless diatoms which would help support primary production. However, toxic dinoflagellates (another species of phytoplankton) thrive on excess nitrogen, so the extra input of nitrogen into the water column may stimulate phytoplankton that could kill fish and other organisms living in the water.
REFERENCES
_
Ball, B., B. Munday, and I. Tuck. "Effects of Otter Trawling on the Benthos and Environment in Muddy Sediments." The Effects of Fishing on Non-target Species and Habitats: Biological, Conservation and Socio-economic Issues. By Michel J. Kaiser and S. J. De Groot. Oxford: Blackwell Science, 2000. 69-82. Print.
Bergman, M. J.N., and J. W. Van Santbrink. "Fishing Mortality of Populations of Megafauna in Sandy Sediments." The Effects of Fishing on Non-target Species and Habitats: Biological, Conservation and Socio-economic Issues. By Michel J. Kaiser and S. J. De Groot. Oxford: Blackwell Science, 2000. 49-68. Print.
Colemon F, Thrush S, Dayton P. 2003. Ecological Effects of Fishing. In Marine Ecosystems of the United States: PEW Oceans Commission. p 1-52.
Ernande B, Dieckmann U, Heino M. 2004. Adaptive changes in harvested populations: plasticity and evolution of age and size at maturation. Proceedings of the Royal Society of London Series B-Biological Sciences 271(1537):415-423.
Garcia SM, Zerbi A, Aliaume C, Chi TD, Lasserre G. 2003. The ecosystem approach to fisheries: Issues, terminology, principles, institutional foundations, implementation and outlook. FAO Fisheries Technical Paper 443:i.
Law R. 2000. Fishing, selection, and phenotypic evolution. Ices Journal of Marine Science 57(3):659-668.
NOAA. Oceanservice. Pressures on Coastal Environments. <oceanservice.noaa.gov/websites/retiredsites/sotc_pdf/IEF.PDF>
Pilskaln, C. , Churchill, J. , & Mayer, L. (1998). Resuspension of sediment by bottom trawling in the gulf of maine and potential geochemical consequences. Conservation Biology, 12(6), 1223-1229.
Resources NCDoEaN. Gag Grouper. Division of Marine Fisheries: N.C. Department of Environment and Natural Resources.
Scott, B. , Marteinsdottir, G. , & Wright, P. (1999). Potential effects of maternal factors on spawning stock-recruitment relationships under varying fishing pressure. Canadian Journal of Fisheries and Aquatic Sciences, 56(10), 1882-1890.
"Shortnose Sturgeon (Acipenser Brevirostrum) - Office of Protected Resources - NOAA Fisheries." Home :: NOAA Fisheries. Ed. Office of Protected Resources. NOAA Marine Fisheries Service. Web. 04 Dec. 2011. <http://www.nmfs.noaa.gov/pr/species/fish/shortnosesturgeon.htm>.
Thrush SF, Dayton PK. 2002. Disturbance to marine benthic habitats by trawling and dredging: Implications for marine biodiversity. Annual Review of Ecology and Systematics 33:449-473.
Thrush SF, Hewitt JE, Funnell GA, Cummings VJ, Ellis J, Schultz D, Talley D, Norkko A. 2001. Fishing disturbance and marine biodiversity: the role of habitat structure in simple soft-sediment systems. Marine Ecology-Progress Series 223:277-286.
Turner SJ, Thrush SF, Hewitt JE, Cummings VJ, Funnell G. 1999. Fishing impacts and the degradation or loss of habitat structure. Fisheries Management and Ecology 6(5):401-420.
Walsh MR, Munch SB, Chiba S, Conover DO. 2006. Maladaptive changes in multiple traits caused by fishing: impediments to population recovery. Ecology Letters 9(2):142-148.
Ball, B., B. Munday, and I. Tuck. "Effects of Otter Trawling on the Benthos and Environment in Muddy Sediments." The Effects of Fishing on Non-target Species and Habitats: Biological, Conservation and Socio-economic Issues. By Michel J. Kaiser and S. J. De Groot. Oxford: Blackwell Science, 2000. 69-82. Print.
Bergman, M. J.N., and J. W. Van Santbrink. "Fishing Mortality of Populations of Megafauna in Sandy Sediments." The Effects of Fishing on Non-target Species and Habitats: Biological, Conservation and Socio-economic Issues. By Michel J. Kaiser and S. J. De Groot. Oxford: Blackwell Science, 2000. 49-68. Print.
Colemon F, Thrush S, Dayton P. 2003. Ecological Effects of Fishing. In Marine Ecosystems of the United States: PEW Oceans Commission. p 1-52.
Ernande B, Dieckmann U, Heino M. 2004. Adaptive changes in harvested populations: plasticity and evolution of age and size at maturation. Proceedings of the Royal Society of London Series B-Biological Sciences 271(1537):415-423.
Garcia SM, Zerbi A, Aliaume C, Chi TD, Lasserre G. 2003. The ecosystem approach to fisheries: Issues, terminology, principles, institutional foundations, implementation and outlook. FAO Fisheries Technical Paper 443:i.
Law R. 2000. Fishing, selection, and phenotypic evolution. Ices Journal of Marine Science 57(3):659-668.
NOAA. Oceanservice. Pressures on Coastal Environments. <oceanservice.noaa.gov/websites/retiredsites/sotc_pdf/IEF.PDF>
Pilskaln, C. , Churchill, J. , & Mayer, L. (1998). Resuspension of sediment by bottom trawling in the gulf of maine and potential geochemical consequences. Conservation Biology, 12(6), 1223-1229.
Resources NCDoEaN. Gag Grouper. Division of Marine Fisheries: N.C. Department of Environment and Natural Resources.
Scott, B. , Marteinsdottir, G. , & Wright, P. (1999). Potential effects of maternal factors on spawning stock-recruitment relationships under varying fishing pressure. Canadian Journal of Fisheries and Aquatic Sciences, 56(10), 1882-1890.
"Shortnose Sturgeon (Acipenser Brevirostrum) - Office of Protected Resources - NOAA Fisheries." Home :: NOAA Fisheries. Ed. Office of Protected Resources. NOAA Marine Fisheries Service. Web. 04 Dec. 2011. <http://www.nmfs.noaa.gov/pr/species/fish/shortnosesturgeon.htm>.
Thrush SF, Dayton PK. 2002. Disturbance to marine benthic habitats by trawling and dredging: Implications for marine biodiversity. Annual Review of Ecology and Systematics 33:449-473.
Thrush SF, Hewitt JE, Funnell GA, Cummings VJ, Ellis J, Schultz D, Talley D, Norkko A. 2001. Fishing disturbance and marine biodiversity: the role of habitat structure in simple soft-sediment systems. Marine Ecology-Progress Series 223:277-286.
Turner SJ, Thrush SF, Hewitt JE, Cummings VJ, Funnell G. 1999. Fishing impacts and the degradation or loss of habitat structure. Fisheries Management and Ecology 6(5):401-420.
Walsh MR, Munch SB, Chiba S, Conover DO. 2006. Maladaptive changes in multiple traits caused by fishing: impediments to population recovery. Ecology Letters 9(2):142-148.